De novo mutations in POGZ are associated with autism spectrum disorder (ASD) and intellectual disability. How disruptions in this gene result in neural circuitry changes that ultimately affect brain function and behavior remain to be determined.
SFARI Investigators Vikaas Sohal and John Rubenstein addressed this question by studying network-level physiology and behavior in Pogz heterozygous mice. This work was supported in part by SFARI Pilot and Research awards.
The researchers found that Pogz+/- mice displayed altered behavior in a well-studied assay of anxiety-related avoidance, the elevated plus maze. Of note, this finding was similar to a recently published study which found decreased anxiety-related behaviors in Pogz mutant mice using a different behavioral assay, the open field test (Matsumura et al., Nat. Commun., 2020).
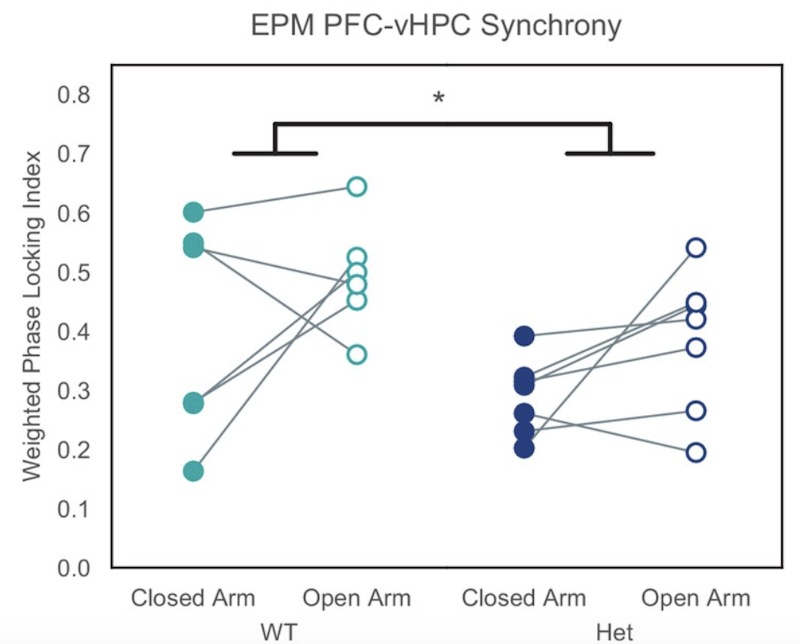
They then studied communication between the ventral hippocampus (vHPC) and medial prefrontal cortex (mPFC), which is known to be necessary for typical anxiety-related avoidance behavior. Theta-frequency synchronization between these two brain regions was decreased in Pogz+/- mice, both at baseline and in the elevated plus maze, compared to wildtype mice. Sohal and Rubenstein’s teams then directly examined vHPC-mPFC connections in brain slices to investigate the physiological changes in more detail. These in vitro studies revealed a loss of excitatory synaptic drive from the vHPC onto prefrontal GABAergic interneurons, whereas excitatory input onto prefrontal pyramidal neurons remained intact. The authors note that this mechanism of impaired feed-forward inhibition could plausibly contribute to the abnormalities that were observed in both avoidance behavior and long-range network connectivity in Pogz+/- mice.
In summary, these findings illustrate how loss of a high-confidence autism gene can impair long-range communication by causing inhibitory circuit dysfunction within pathways important for specific behaviors. Such knowledge is an important first step toward understanding which circuits in the developing brain might be targeted to address behavior.
Reference(s)
Altered hippocampal-prefrontal communication during anxiety-related avoidance in mice deficient for the autism-associated gene Pogz.
Cunniff M.M., Markenscoff-Papadimitriou E., Ostrowski J., Rubenstein J., Sohal V.


