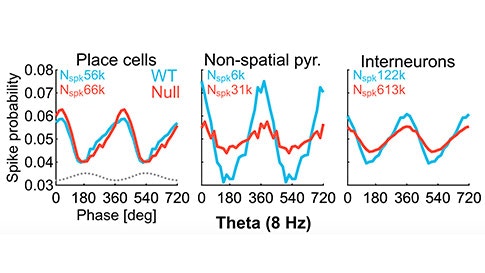
Fragile X syndrome (FXS) is an inherited form of intellectual disability often associated with autism spectrum disorder (ASD), which results from the silencing of the fragile X mental retardation gene 1 (FMR1). Electrophysiological studies have demonstrated dysfunction in synaptic communication in Fmr1-null mice and other models of ASD, leading to the hypothesis that synaptic pathophysiology is involved in mediating ASD-linked behaviors. However, Fmr1-null mice also show larger-scale network-level disruptions in neuronal communication. To tease apart the mechanisms mediating ASD-linked behavioral alterations, SFARI Investigator André Fenton and colleagues assessed synaptic- and network-level neuronal processing in Fmr1-null mice performing a spatial-location task. In doing so, Fenton et al. demonstrated that hippocampal neurons in Fmr1-null mice show normal activation in response to spatial location, arguing that synaptic disruptions are not occurring during this behavior. However, Fmr1-null mice did show discoordination in hippocampal network firing that was predictive of poorer cognitive performance in place-avoidance training. Combined, these data suggest larger-scale alterations in the coordination of neuronal network communication, in addition to defects in communication between individual hippocampal neurons, may play a role in ASD, including FXS.
Reference(s)
Normal CA1 place fields but discoordinated network discharge in a Fmr1-null mouse model of fragile X syndrome.
Talbot, Z.N., Sparks, F.T., Dvorak, D., Curran, B.M., Alarcon, J.M., Fenton A.


