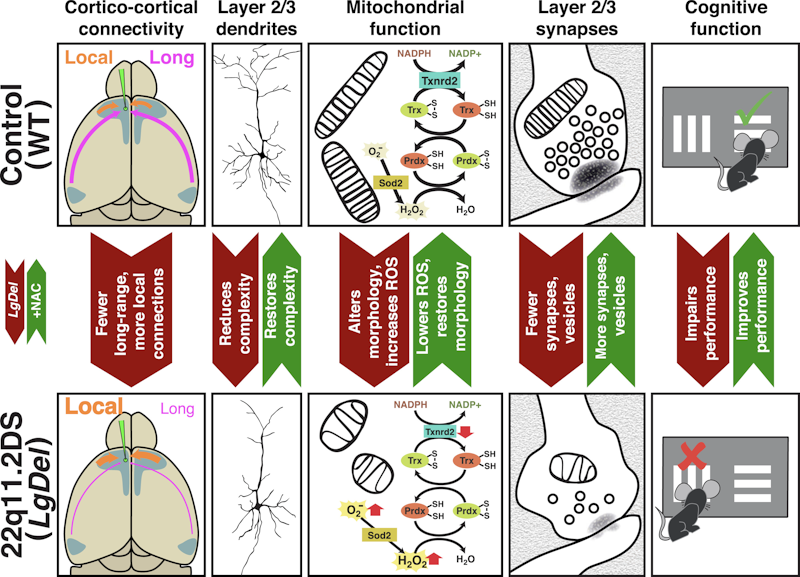
Deletions at the 22q11.2 locus are known to be strong risk factors for schizophrenia and autism spectrum disorder (ASD), with several genes in the interval having been identified as contributing to different aspects of the associated phenotype. Building on previous work implicating dysfunctional mitochondria in 22q11.2 syndrome (Meechan et al., Int. J. Neurosci., 2011), SFARI Investigators Anthony-Samuel LaMantia and Thomas Maynard and colleagues now define the impact of mitochondrial dysfunction on long-range cortico-cortical connectivity in this disorder.
In the new work, supported in part by a SFARI Research Award, LaMantia’s team showed that there is increased local connectivity and reduced long-range connectivity in a mouse model of 22q11.2 deletion and that this is accompanied by a range of cellular effects that seem to be restricted to upper-layer (layer 2/3) neurons. Specifically, they observe layer 2/3 axons and dendrites that are shorter and have fewer branches, and harbor mitochondria that are both fewer in number and show increases in reactive oxygen species (ROS).
They provide additional evidence that this mitochondrial defect is driven by Txnrd2, a gene encoding a key mitochondrial enzyme involved in the clearance of ROS. Moreover, administration of antioxidants such as N-acetyl cysteine improves mitochondrial, synaptic, dendritic and cognitive behavioral measures in the mice. LaMantia and colleagues argue that long-distance cortico-cortical connections may be more sensitive to subtle disruptions in mitochondrial function given the greater bioenergetic demands involved in generating these projections.
The particular focus on layer 2/3 neurons is of note in light of other studies that have also implicated them in ASD pathophysiology (Parikshak et al., Cell, 2013; Velmeshev et al., Science, 2019). Finally, we note that genetic evidence for disruptions in mitochondrial function in ASD (Wang et al., PLoS Genet., 2016) continues to accumulate.
Reference(s)
Mitochondrial dysfunction leads to cortical under-connectivity and cognitive impairment.
Fernandez A., Meechan D.W., Karpinski B.A., Paronett E.M., Bryan C.A., Rutz H.L., Radin E.A., Lubin N., Bonner E.R., Popratiloff A., Rothblat L.A., Maynard T., LaMantia A.


