Ethan Greenblatt, Ph.D.
Assistant Professor, University of British Columbia
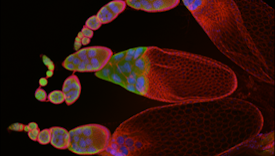
SFARI Investigator WebsiteEthan Greenblatt’s laboratory is interested in the biology of translational regulation in disease and aging, with a focus on the role of RNA-binding proteins in controlling gene expression. One of its main focuses is on the FMR1 gene, which is mutated in fragile X syndrome and fragile X–associated primary ovarian insufficiency, prominent heritable causes of intellectual disability, autism spectrum disorder and premature infertility. Greenblatt’s lab uses a novel model of translational control, the mature Drosophila oocyte. The Drosophila oocyte is an advantageous model in which to study translational regulation because it is a large, transcriptionally silent cell which relies entirely on the ongoing translational control of stored mRNAs.
Greenblatt received his B.S. in chemistry from the College of William and Mary. He then performed doctoral studies in biophysics in Ron Kopito’s lab at Stanford University. During this time, he worked on the endoplasmic reticulum-associated degradation (ERAD) pathway and identified a central role for membrane rhomboid pseudoproteases in the trafficking of misfolded secretory proteins to the cytoplasm en route to proteasomal destruction.
Greenblatt then joined Allan Spradling’s lab at the Carnegie Institution for Science for his postdoctoral fellowship, where he established a novel system for studying the biology of developmental arrest using quiescent mature Drosophila oocytes. This led to the identification of a key role for aberrant translation in the loss of oocyte function during normal aging or in the absence of disease-associated genes such as FMR1.
The long-term goal of Greenblatt’s laboratory is to understand the mechanism of FMR1-dependent translation and to further elucidate the role of translation dysfunction in meiotic errors associated with oocyte aging.