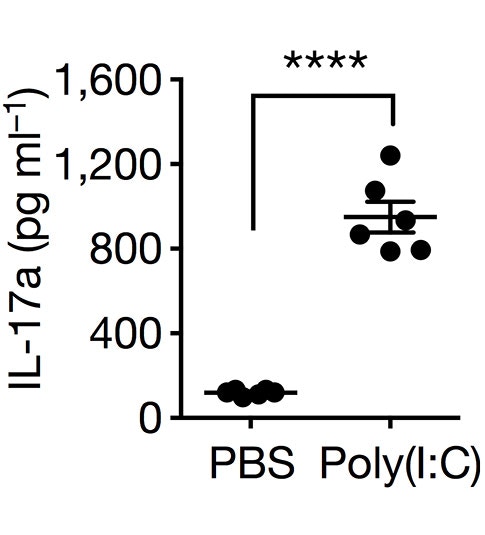
Epidemiological studies in humans, and related studies in animal model systems, have provided evidence that maternal infection, and the subsequent inflammatory response during pregnancy, leads to an increased risk of the fetus developing autism-related behaviors. It has been suggested that the inflammatory factor interlukin-17a (IL-17a) plays a role, but the biological mechanisms that underlie maternal immune activation (MIA)-induced behavioral alterations have remained unclear. In two papers recently published in Nature, SFARI Investigators Jun Huh and Gloria Choi, and their collaborators including SFARI Investigator Dan Littman, asked whether the intestinal microbiota also plays a role in MIA-induced behaviors. Huh’s laboratory reports that infection in pregnant female mice causes a specific type of gut microbiota, called ‘segmented filamentous bacteria,’ to increase differentiation of T helper 17 cells (TH17), the cells that produce IL-17a. Infection in nonpregnant female mice does not affect TH17 differentiation, nor is this effect observed in pregnant female mice lacking the segmented filamentous bacteria (SFB). Expanding on these findings, Choi’s laboratory finds that MIA has a specific effect on the development of the primary somatosensory cortex in pups born to mothers carrying the SFB. The authors describe cortical patches in a dysgranular portion of this area (S1DZ), the size of which correlates with behavioral abnormalities. By using optogenetic techniques, Choi’s group also shows that MIA-induced behaviors are associated with neuronal excitatory/inhibitory imbalance in this area, as well as altered connectivity with motor, temporal and striatal regions. Combined, these findings offer key insights into the pathophysiological mechanisms underlying MIA-induced behavioral alterations and provide a first step toward understanding how the risk of neurodevelopmental disorders associated with maternal inflammation may be reduced and therapeutic interventions eventually developed.
Reference(s)
Maternal gut bacteria promote neurodevelopmental abnormalities in mouse offspring.
Kim S., Kim H., Yim Y. S., Ha S., Atarashi K., Tan T.G., Longman R.S., Honda K., Littman D., Choi G. B., Huh J.


