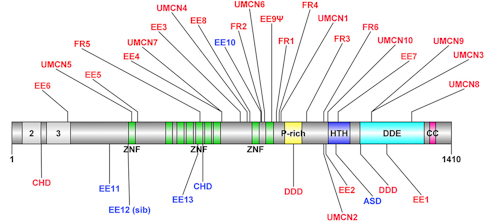
A small number of mutations in the Pogo transposable element with zinc finger domain (POGZ) have been linked to autism spectrum disorder (ASD) and intellectual disability. Yet due to a rarity of occurrence in the population, clarifying the pathogenicity of POGZ mutations and their behavioral consequences has proven difficult. Using whole-exome, whole-genome and targeted sequencing of multiple cohorts of individuals with neurodevelopmental disorders, SFARI Investigator Evan Eichler and his collaborators gathered a cohort of 25 individuals with de novo mutations in POGZ. Detailed phenotypic assessments of these individuals enabled Eichler’s team to define a core phenotype associated with mutations in POGZ. By comparing the phenotypes of POGZ individuals diagnosed with ASD with phenotypic data previously gathered from the Simons Simplex Collection (SSC), the team found that individuals with POGZ mutations represent a distinct clinical ASD subtype. Continued characterization of ‘genetics-first’ cohorts will help to further refine our understanding of how the diverse range of clinical and behavioral phenotypes associated with ASD relate to genetic heterogeneity.
Reference(s)
Disruption of POGZ is associated with intellectual disability and autism spectrum disorders.
Stessman H., Willemsen M.H., Fenckova M., Penn O., Hoischen A., Xiong B., Wang T., Hoekzema K., Vives L., Vogel I., Brunner H.G., van der Burgt I., Ockeloen C.W., Schuurs-Hoeijmakers J.H., Klein Wassink-Ruiter J.S., Stumpel C., Stevens S.J., Vles H.S., Marcelis C.M., van Bokhoven H., Cantagrel V., Colleaux L., Nicouleau M., Lyonnet S., Bernier R., Gerdts J., Coe B.P., Romano C., Alberti A., Grillo L., Scuderi C., Nordenskjöld M., Kvarnung M., Guo H., Xia K., Piton A., Gerard B., Genevieve D., Delobel B., Lehalle D., Perrin L., Prieur F., Thevenon J., Gecz J., Shaw M., Pfundt R., Keren B., Jacquette A., Schenck A., Eichler E., Kleefstra T.


