Dysregulated levels of neuromodulators and other chemical signals may contribute to behavioral characteristics of ASD. Yet previous efforts have often focused on only one signal at a time, and typically provide a static description of signal levels in the brain. In the current project, Mark Andermann and colleagues plan to use novel optical methods to track and control dozens of neuromodulators and peptides in the brain of a genetic mouse model of ASD.

On August 4, 2022, SFARI and Simons Searchlight helped to host two scientific workshops focused on neurodevelopmental disorders (NDDs) that are linked to mutations in the CSNK2A1 and SETBP1 genes. Meeting in Baltimore, the workshops were held in conjunction with a family conference that gathered together people with these conditions and their family members.

Seizures are an extreme outcome of excitatory-inhibitory imbalance and are the most common neurological complication in autism spectrum disorder (ASD). Seizures are even more common in syndromic forms of ASD such as Angelman syndrome. In the current project, Ben Philpot’s laboratory aims to identify the circuitry and protein pathways underlying seizures in a mouse model of Angelman syndrome, with the goal of identifying disease-modifying targets to treat seizures. The mechanistic insights yielded by these studies may further guide therapeutically oriented investigations of excitatory-inhibitory imbalance across the broader spectrum of ASDs.

The sensory and neural mechanisms that mediate social communication facilitating attachment and how they are affected in the context of autism spectrum disorder (ASD) are poorly understood. Prairie voles are small rodents that display long-term relationships between peers and mates. Devanand Manoli proposes to understand how specific mutations in two ASD risk genes, Shank3 and Scn2a, disrupt the processing of social cues, leading to the identification of brain regions that could inform targeted interventions to improve social communication in ASD.
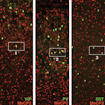
Jessica Cardin and colleagues identified an important and unexpected role for the VIP subclass of interneurons in mediating the functions of MeCP2 in the development of cortical circuits.

Emmanuel Mignot discussed sleep biology as well as sleep disorders and their impact. He presented a link to what is known on the genetics of sleep and sleep disorders. He emphasized the need for large scale objective sleep recording studies with genomic and proteomic analysis to better understand the molecular pathways regulating sleep and circadian biology.

Emmanuel Mignot is the Craig Reynolds Professor of Sleep Medicine at Stanford University. He discovered that human narcolepsy is caused by an autoimmune loss of approximately 20,000 hypothalamic neurons secreting the wake-promoting peptide hypocretin (also known as orexin). He also identified HLA-DQB1*06:02 and T-cell receptor genes as major susceptibility genes, which act together to promote a selective autoimmune process triggered by influenza A. Mignot has received numerous awards and is a member of the National Academy of Sciences and the National Academy of Medicine.

On May 21, 2019, the recipients of the SFARI Bridge to Independence Award gathered at the Simons Foundation to discuss their scientific findings and plans in autism research.
On April 17, 2018, molecular neuroscientists and geneticists gathered at the Simons Foundation for a workshop on the role of chromatin-associated proteins in autism spectrum disorder. The workshop discussed the biology and function of these proteins in brain development, while considering translational opportunities and evaluating ways that SFARI could potentially help move research in this area forward.
- Previous Page
- Viewing
- Next Page