SFARI is pleased to announce that it intends to fund 19 grants in response to the Summer 2020 Pilot Award request for applications.

SFARI is pleased to announce that it intends to fund 19 grants in response to the Summer 2020 Pilot Award request for applications.

SYNGAP1 encodes a neuronal Ras GTPase activating protein and is a significant risk gene associated with autism spectrum disorders (ASDs) and intellectual disability (ID). As many of the genetic mutations in individuals with SYNGAP1-related ID (SRID) lead to decreased SYNGAP1 expression, SRID is an ideal candidate for genetic and antisense oligonucleotide–based therapies that increase SYNGAP1 expression. Leveraging recently discovered regulatory mechanisms of SYNGAP1 expression, Richard Huganir’s team plans to design precision antisense oligonucleotides that increase SYNGAP1 expression and to validate them using human pluripotent stem cell models of SRID. These studies will help to advance the therapeutic potential of antisense oligonucleotide–based treatments for SRID as well as other monogenic forms of ID and ASD.
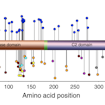
Kurt Haas and colleagues used multiple models and bioassays to assess the functional impact of more than 100 missense and nonsense mutations in PTEN, allowing for high-confidence predictions of pathogenicity.

New Simons Searchlight data were recently added to SFARI Base. This data release included phenotypic data from individuals with 16p11.2 copy number variants (CNVs), 1q21.1 CNVs, 7q11.23 duplication and variants in 29 single genes associated with autism and related neurodevelopmental conditions.

Gastrointestinal (GI) distress commonly accompanies autism spectrum disorders (ASDs), significantly impacting the quality of life of those affected and their families. Julia Dallman, in collaboration with John Rawls, plans to use zebrafish as an experimental system, since it allows for the GI tract to be imaged and manipulated in live animals. They aim to determine if GI phenotypes in multiple genetic forms of ASD are caused by convergent gut-intrinsic mechanisms. The expected outcomes would open a new field of GI research for ASD that could suggest treatment strategies for managing GI distress in humans.
A new collaboration between SFARI and the Nancy Lurie Marks Family Foundation will generate hundreds of induced pluripotent stem cells from individuals with autism and related neurodevelopmental conditions. These cell lines will become available to researchers starting next year.

Shinjae Chung and Ted Abel will assess the neural dynamics of sleep neurons in Syngap1 mutant mice, aiming to understand how changes in their activity lead to sleep disturbances and behaviors associated with autism.
SFARI is now curating a set of zebrafish lines to study autism spectrum disorder. This includes mutant lines for 12 ASD risk genes, four of which are currently available to researchers and eight that will be available later this year.

SFARI is helping to make zebrafish models of high-risk autism genes available to the research community.