
New phenotypic data from Simons Searchlight participants were recently added to SFARI Base. This release includes data from individuals with 59 gene changes and nine copy number variants known to be connected to autism.

New phenotypic data from Simons Searchlight participants were recently added to SFARI Base. This release includes data from individuals with 59 gene changes and nine copy number variants known to be connected to autism.
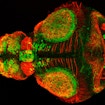
New zebrafish lines with mutations in the high-confidence autism risk genes DYRK1A and GRIN2B have been added to SFARI resources.

New Simons Searchlight data were recently added to SFARI Base. The data released included phenotypic data from individuals with 16p11.2 copy number variant (CNVs), 1q21.1 CNVs, 7q11.23 duplication and variants in 32 single genes associated with autism and related neurodevelopmental conditions.

New phenotypic data from Simons Searchlight participants were recently added to SFARI Base. This release includes data from individuals with 94 gene changes and 15 copy number variants known to be connected to autism.

SFARI is pleased to announce it will fund five projects investigating the influence of autism-associated risk genes on early neurodevelopment and links to later circuit-level functional outcomes. These awards will provide up to $3.6 million over 4 years to each team of researchers.

On September 29, 2025, the SFARI Autism Rat Consortium (ARC) held its annual meeting in the Usher Institute of the University of Edinburgh. This year’s event brought together ARC PIs and trainees who specialize in the use of rat models.

Based on the critical role of silent synapses in developmental neurocircuit refinement, Oliver Schlüter aims to assess whether ASD-risk genes encoding proteins associated with glutamate receptor complexes play a common role in silent synapse development. Using three different ASD mouse models (Shank3, Syngap1 and Nlgn3 deficiency), Schlüter will assess whether alterations in silent synapse maturation represent a common mechanistic defect underlying the distinct phenotypic facets of ASDs.

In the current project, Arpiar Saunders and his lab plan to determine how variants in the ASD risk genes GRIN2B and SYNGAP1 alter molecular and synaptic properties of mouse somatosensory cortical circuits. To achieve this goal, they will use next-generation viral tools and high-throughput single-cell RNA sequencing that enable highly parallelized connectivity and molecular phenotyping of mouse cells expressing human alleles in the intact brain.

Bateup will use genetic mouse models of ASD to investigate the idea that synaptic alterations in the striatum are central to the inflexible behaviors observed in ASD.

Jess Cardin and Michael J. Higley will establish a functional screen (using a CRISPR/Cas9-induced gene disruption system and multiscale in vivo calcium imaging in awake mice) for the assessment of common cellular- and circuit-level cortical dysregulation phenotypes associated with mutations in ASD risk genes.